Temperature conditions in sewers are moderate and almost always in a range to promote biological activity. They can exist at temperatures ranging from 32°F to 212°F with the optimum range being 75°F to 107°F. Many factors affect this activity including; design capacity versus actual flows, retention time, turbulence, organic waste levels, biological oxygen demand levels, industrial pollutants (known to impede biological activity) and Inflow and Infiltration (I & I.) Efforts to reduce industrial pollutants and I & I in many cases have increased the biological activity. Reducing I & I increases retention times as well as concentrates the organic waste levels. These efforts, while needed for many reasons, are causing increased MIC in the sewers.
The first step:

This is a complex chain of events that begins with anaerobic (non-air breathing) bacteria thriving in the wastewater stream and in slim layers (again under the water) attached to structures of all types. They are naturally abundant and impossible to prevent naturally. There are estimates that certain human waste types may be comprised of ~ 54% bacteria cells (including dead and lives ones and both aerobic and anaerobic types.) The raw sewage may contain as many as 1 x 1013 cells per milliliter. There are many kinds including those that feed on compounds of sulphur, nitrogen and phosphorus, etc. In the case of MIC, it is the sulphur-reducing strains that consume the many sulphur compounds in the water that concern us. Sulfates are found in drinking water (10 to 50 ppm) and food and in turn human waste. They can be found in raw sewers up to 8,000 ppm. In a typical 1 Million Gallon per Day (MGD) sewer, a typical value could be 20 tons of sulfates flowing each day.
The second step:

The anaerobic bacteria reduce the sulfates and produce various sulphides. One of them is hydrogen sulphide (H2S), a gas slightly heavier than air. (This is a great over-simplification and more details can be found on this topic further in the paper when chemical additions are discussed.) The solubility of H2S in water is a function of temperature and pH but generally it is around 3,850 ppm. This is significantly higher (by ~ 100 times) than hydrogen or oxygen or nitrogen and about half that of Chlorine and twice that of CO2. Nevertheless, H2S is easily separated from water with simple aeration. With the high concentrations it can also simply boil out of solution with just limited movement of the water like that of a typical gravity sewer or force main.
The third step:
With an abundant amount of H2S gas in the moist oxygen rich atmosphere above the liquid level, the second important biological activity takes place. Here aerobic (air breathing) Thiobacillus bacteria eventually thrive. They rise from the liquid and collect on all surfaces. These bacteria consume the H2S gas along with water vapor and oxygen via a complex process in itself, and secrete dilute sulphuric acid as their waste product. They do not need anything further to survive. In a new concrete structure, the surface alkalinity is high with a pH generally greater than 11. Over time, and again many factors can affect this timing, H2S and carbonation along with other general atmospheric conditions reduce the alkalinity of new concrete surfaces. In the case of PVC pipe or various coatings, the bacteria can thrive much more quickly as there is no alkalinity on the surfaces. The bacteria have a neutral surface on which to begin the colonization process. With concrete surfaces, the process is complex and involves the evolution of at least 5 strains of the sulphur consuming Gram-negative bacteria. The first strains can tolerate surface pHs as high as 9 and produce only modest amounts of very weak acid. But they are important as they work to lower the surface pH to where more prolific bacteria thrive and produce stronger acids capable of quickly destroying concrete. Some species of Thiobacillus thrive in sulfuric acid solutions as concentrated as 7 percent with a pH approaching zero. They are prolific producers of acid. Keep in mind the pH scale is logarithmic and a concentration of just 0.03% results in a pH of ~ 2.
The fourth step:
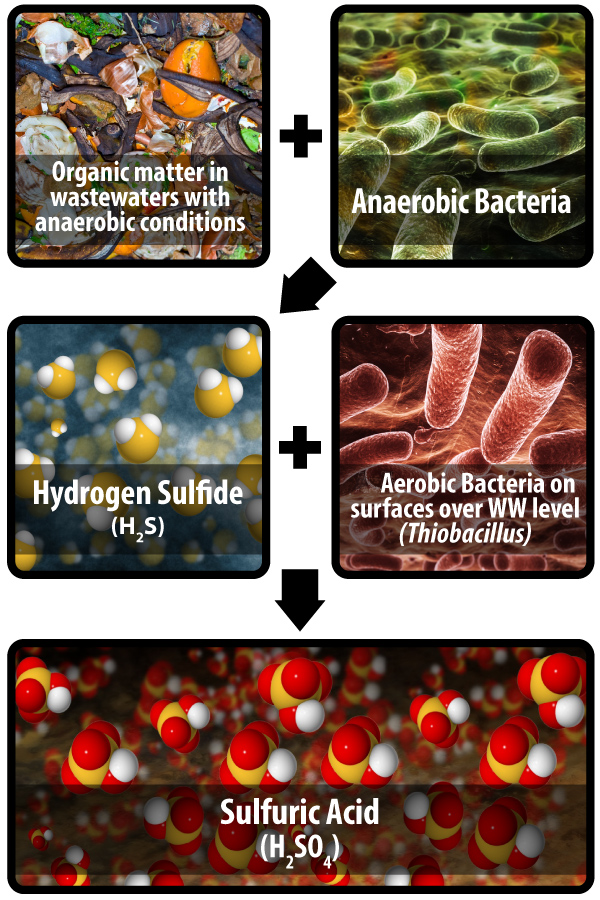
The destruction of the concrete at this point is a simple matter of dissolving the various cement hydrates. This will be explained in detail below. What is left behind is a crumbly muck of aggregates and calcium sulfate complexes with no structural value. No means exist to render normal concretes capable of directly resisting this chemical attack. The only rational solutions include stopping the production of the acid or by trying to prevent the acid from coming in contact with the concrete.
MIC, once firmly established, can destroy concrete integrity in a few years.
Much is often said of designing sewers to minimize MIC and odors. There are several complex computer models that attempt to predict odor and corrosion rates but they require many factors to be known. Some parameters are raised to powers such as 3/8. This seems most exacting when one considers it is difficult to determine linear or even a 2nd order equation in nature. They work somewhat for understanding existing sewers but not so well for forecasting the behavior in new construction. There is not much that can be done from a simple design point of view. Sewers are already very simple structures. As discussed there is a tremendous and complex biological machine involved.
As an example, a 36” concrete sanitary sewer was inspected ~ 6 years ago as part of a routine 5 year process. It was found to be in very good condition. Subsequently, the utility went on an extensive program to reduce I & I. The program was very successful and when the sewer was again inspected in ~ 2016 it was found to be in terrible condition. In areas the crown was completely gone. The reduction in I & I reduced flows and increased retention time. Also heavy rains were previously helping to occasionally scour the bio mass from the invert.



